Gregory A. Marus, Eduardo Vyhmeister, Pamela Pollet, Megan E. Donaldson, Veronica Llopis-Mestre, Sean Faltermeier, Renee Roesel, Leslie Gelbaum, Charles L. Liotta, and Charles A. Eckert, “The Sustainable and Scalable Synthesis of Piperylene Sulfone: A “Volatile” and Recyclable DMSO Substitute,” I&EC Res, 50, 23-27, 2011.
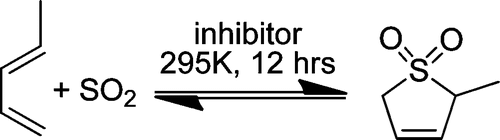
An essential feature of chemical research is advancing new laboratory findings to a form useful to industry. Dimethyl sulfoxide (DMSO) is an important dipolar, aprotic solvent for conducting chemical reactions. Unfortunately, separation of the products of reaction from the solvent is difficult and expensive. We have proposed the use of piperylene sulfone (PS) as a substitute for DMSO, and herein, we establish a roadmap to its sustainable and scalable synthesis. PS is a potentially important new dipolar aprotic solvent that has solvent properties similar to those of DMSO; in contrast to DMSO, PS is fully recyclable and undergoes a reversible retrocheletropic reaction at 110 °C, permitting facile solvent removal and recycle. PS is synthesized by the reaction of trans-piperylene with sulfur dioxide. Because PS is not commercially available, we synthesized laboratory quantities using a method not sustainable on a large scale because of expensive chemicals and considerable waste generation. To develop and optimize a scalable process, we (1) determined the kinetic parameters associated with the reaction by employing in situ proton NMR measurements and (2) studied the effects of radical inhibitors in reducing unwanted side reactions. In addition, we recovered PS from the reaction mixture through a sustainable CO2 separation method, which resulted in a substantial waste reduction. Our development of a more efficient, safe, and sustainable scaleup method for PS thus illustrates an important aspect of chemical research: the need to render the results usable and useful to industry.
Ali Z. Fadhel, Pamela Pollet, Charles L. Liotta, Charles A. Eckert, “Novel Solvents for Sustainable Production of Specialty Chemicals,” Annual Review of Chemical & Biomolecular Engineering, 2, 189-210, 2011.
We discuss novel solvents that improve the sustainability of various chemical reactions and processes. These alternative solvents include organic-aqueous tunable solvents; near-critical water; switchable piperylene sulfone, a volatile dimethylsulfoxide substitute; and reversible ionic liquids. These solvents are advantageous to a wide variety of reactions because they reduce waste and energy demand by coupling homogeneous reactions with heterogeneous separations, acting as in situ acid or base catalysts, and providing simple and efficient postreaction separations.
Ali Z. Fadhel, Wilmarie Medina-Ramos, Andy Wu, Jackson Ford, Veronica Llopis-Mestre, Rani Jha, Pamela Pollet, Charles L. Liotta, Charles A. Eckert, “Exploiting Phase Behavior for Coupling Homogeneous Reactions with Heterogeneous Separations in Sustainable Production of Pharmaceuticals,” JChem Eng Data, 56, 1311-1315, 2011.
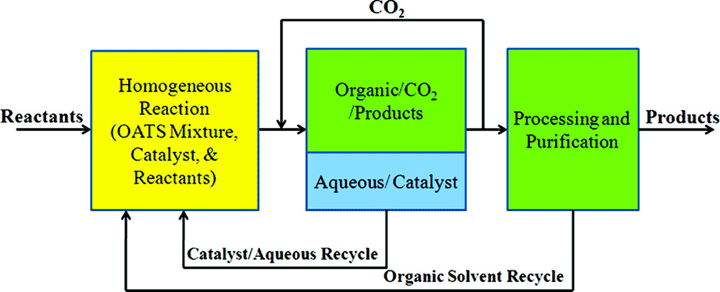
John Prausnitz was never content to measure new data solely because the data were new. He always had an application in mind—a separation or even a process that required the data for implementation. We present here an advance in using designed changes in phase equilibria to enable the facile recovery and recycle of homogeneous catalysts. We show a new application of organic-aqueous tunable solvents (OATS) to run homogeneous reactions (fast rates and high selectivities) followed by facile and efficient heterogeneous separations and the recycle of the homogeneous catalysts. This is done by using CO2 to manipulate the phase behavior of monophasic organic-water mixtures to form heterogeneous organic-rich and aqueous-rich phases. The example shown is the hydroformylation of hydrophobic p-methylstyrene catalyzed by rhodium catalyst to which is attached a hydrophilic phosphorus ligand. The OATS method increases the conversion rate of styrene to the aldehyde products by an order of magnitude compared to heterogeneously reported reactions. Also, the selectivity toward the branched aldehyde (the desired product) increases by 30 %. The hydrophobic product partitions into the organic-rich phase with more than 99 % removal efficiency, and the hydrophilic catalyst is retained in the aqueous-rich phase with 99.9 % efficiency. In addition, we recycle the catalyst for five consecutive reactions without significant loss of catalytic activity.
Pamela Pollet, Charles A. Eckert, and Charles L. Liotta, “Switchable Solvents,” Chem. Sci, 2, 609 – 614, 2011
 . .
Switchable solvents are a unique class of solvents that were developed to facilitate both reaction and subsequent product separation. Their ‘‘built-in’’ separation ability for facile product recovery is paramount to achieving chemical processes that are both economically competitive and environmentally conscious. Two classes of switchable solvents are discussed: 1) piperylene sulfone— a volatile and recycle DMSO substitute and 2) one and two-component reversible ionic liquids— solvent systems that can be switched back and forth between molecular liquids and ionic liquids.
P. Pollet, C. L. Liotta, C. A. Eckert, M. Verma, E. Nixon, S. Sivaswamy, R. Jha, F. Momin, L. Gelbaum, B. I. Chaudhary, J. M. Cogen, “Radical-Mediated Graft Modification of Polyethylene Models with Vinyltrimethoxysilane: A Fundamental Study”, Ind. Eng. Chem. Res., 50, 12246–12253, 2011.
Cross-linked polyethylenes have widespread applications such as insulators for current-carrying wires/cables and for hot water pipes. Industrially, cross-linked polyethylene can be prepared via the radical-initiated grafting of polyethylene with vinyltrimethoxysilane (VTMS) followed by moisture-induced cross-linking. The efficiency of the grafting reaction is not optimal: (1) a large portion of the VTMS remains unreacted and (2) controlling the distribution and extent of grafting is difficult. We present new contributions to the fundamental understanding of grafting reactions with VTMS on polymeric models (dodecane and heptane) as to the effect of phenyllithium capping reaction time on the analyses, the origin of the observation of multiple grafts, and the regiochemistry of the multiple graft products.
Amy Rohan, Jackson Switzer, Kyle Flack, ryan Hart, Swetha Sivaswamy, Elizabeth Biddinger, Manish Talreja, Manjusha Verma, Sean Faltermeier, Paul Nielsen, Pamela Pollet, George Schuette, Charles Eckert, Charles Liotta, “The Synthesis and the Chemical and Physical Properties of Non-Aqueous Silylamine Solvents for CO2 Capture,” ChemSusChem, 5, 11, 2181-2187, 2012.

Silylamine reversible ionic liquids were designed to achieve specific physical properties in order to address effective CO2 capture. The reversible ionic liquid systems reported herein represent a class of switchable solvents where a relatively non-polar silylamine (molecular liquid) is reversibly transformed to a reversible ionic liquid (RevIL) by reaction with CO2 (chemisorption). The RevILs can further capture additional CO2 through physical absorption (physisorption). The effects of changes in structure on (1) the CO2 capture capacity (chemisorption and physisorption), (2) the viscosity of the solvent systems at partial and total conversion to the ionic liquid state, (3) the energy required for reversing the CO2 capture process, and (4) the ability to recycle the solvents systems are reported.
Kyle Flack,Kristen Kitagawa,Pamela Pollet,Charles A. Eckert,Kent Richman, Joy Stringer, William Dubay, and Charles L. Liotta, “Al(OtBu)3 as an Effective Catalyst for the Enhancement of Meerwein-Ponndorf−Verley (MPV) Reductions,” Organic Process Research & Development, 16, 1301-1306, June2012.

The Meerwein–Ponndorf–Verley (MPV) reduction of aldehydes and ketones has been the cornerstone in many multistep syntheses. Herein we report the use of Al(OtBu)3 instead of the commonly used Al(OiPr)3 which results in a dramatic rate increase and significantly lower catalyst loading for the reduction of (1) model compounds benzaldehyde and acetophenone, and (2) N-(tert-butyloxycarbonyl)-(3S)-3-amino-1-chloro-4-phenyl-2-butanone or (S)-CMK, a key intermediate in HIV protease inhibitor synthesis.
Maria Gonzalez-Miquel, Manish Talreja, Amy Ethier, Kyle Flack, Jackson Switzer, Elizabeth Biddinger, Pamela Pollet, Jose Palomar, Francisco Rodriguez, Charles Eckert, and Charles Liotta, “COSMO-RS Studies: Structure-property Relationships for CO2 Capture by Reversible Ionic Liquids,” I&EC Res, 51, 16066-16073, 2012.
The quantum-chemical approach COSMO-RS was used to develop structure–property relationships of reversible ionic-liquid (RevIL) solvents for CO2 capture. Trends predicted for the thermodynamic properties of the RevILs using COSMO-RS, such as CO2 solubility, solvent regeneration enthalpy, and solvent reversal temperature, were verified by experimental data. This method was applied to a range of structures, including silylamines with varying alkyl chain lengths attached to the silicon and amine functionality, silylamines with fluorinated alkyl chains, sterically hindered silylamines and carbon-based analogues. The energetics of CO2 capture and release and the CO2 capture capacities are compared to those of the conventional capture solvent monoethanolamine. The results of this study suggest that the simple COSMO-RS computational approaches reported herein can act as a guide for designing new RevILs. COSMO-RS allows for the determination of the relative thermodynamic properties of CO2 in these and related systems.
|
